measure 20ml precisely chemistry, a activity that appears easy however may be difficult if not carried out appropriately. Measuring quantity precisely is essential in laboratory procedures, and one of the crucial frequent measurements is 20ml. It isn’t nearly following a process, but additionally about understanding the significance of precision and accuracy in your work.
On this information, we’ll take you thru the significance of correct measurement in chemistry, frequent strategies for measuring volumes, greatest practices for measuring 20ml precisely, and extra. We’ll cowl the challenges it’s possible you’ll face and offer you tips about methods to overcome them. By the top of this text, it is possible for you to to measure 20ml precisely and confidently.
Significance of Correct Measurement in Chemistry
Correct measurement is the cornerstone of any scientific experiment, particularly in chemistry. The precision and reliability of measurements instantly impression the experimental outcomes, which in flip inform our understanding of chemical processes and reactions. Inaccurate measurements can result in deceptive or invalid outcomes, undermining the credibility of scientific analysis.
One of many major causes for the significance of correct measurement in chemistry is the numerous impression it has on experimental outcomes. Even a small discrepancy in measurements can lead to a profound distinction within the experimental outcomes. That is very true in chemistry, the place the legal guidelines of stoichiometry and the conduct of chemical compounds are finely tuned. Small variations in measurements can alter the proportions of reactants and merchandise, resulting in incorrect conclusions about chemical reactions and processes.
Listed here are three key eventualities the place correct measurement is critically essential in chemistry:
Conditions The place Inaccurate Measurements Can Result in Deceptive Outcomes
Chemical reactions typically depend on exact quantitative relationships between reactants and merchandise. Inaccurate measurements of reactants can result in incorrect conclusions concerning the response’s equilibrium fixed, response charge, and general yield.
- As an example, in a titration experiment, inaccurate measurements of the titrant’s quantity can lead to an incorrect calculation of the analyte’s focus, resulting in a mistaken conclusion concerning the chemical’s properties.
- In a gasoline legislation experiment, exact measurements of temperature and strain are essential. Inaccurate measurements can result in an incorrect interpretation of the conduct of gases beneath completely different circumstances.
- Moreover, the accuracy of measurements is paramount in spectroscopic evaluation, the place the evaluation of absorption and emission spectra depends on exact quantification of wavelength and absorbance.
Strategies for Measuring Volumes Precisely
Chemical laboratories make use of varied methods to make sure correct quantity measurements. Among the commonest strategies embrace:
- Burettes: Burettes are exact glass vessels with graduated markings that permit for correct measurement of small volumes (1-50 mL). They’re generally used for measuring dilute acids and bases.
- Microburettes: These are small, exact glass vessels used for measuring tiny volumes (0.1-1 mL). They are perfect for dealing with diluted options.
- Measuring cylinders: These are glass vessels with graduated markings that permit for correct measurement of bigger volumes (10-1000 mL). They’re generally used for transferring and mixing reagents.
Evaluating Completely different Strategies for Correct Quantity Measurement
The selection of methodology depends upon the precise necessities of the experiment and the properties of the chemical being measured. Some strategies are higher suited to sure functions than others:
| Technique | Accuracy | Vary | Purposes |
|---|---|---|---|
| Burettes | ±0.1 mL | 1-50 mL | Dilute acids and bases, titration experiments |
| Microburettes | ±0.01 mL | 0.1-1 mL | Dealing with diluted options, spectrophotometry |
| Measuring cylinders | ±1 mL | 10-1000 mL | Transferring and mixing reagents |
Significance of Accuracy in Measurement for Predicting Conduct of Chemical substances
The accuracy of measurements is paramount for predicting the conduct of chemical compounds and understanding chemical processes. Inaccurate measurements can result in incorrect conclusions concerning the properties and conduct of chemical compounds, undermining the reliability of scientific analysis and its functions in on a regular basis life.
The significance of correct measurement in chemistry can’t be overstated. Precision and reliability are important for understanding chemical processes, predicting chemical conduct, and informing decision-making in varied fields. By using correct measurement methods and punctiliously deciding on probably the most appropriate methodology for the duty at hand, researchers can make sure the validity and reliability of their outcomes, resulting in a deeper understanding of the complicated world of chemistry.
Frequent Strategies for Measuring Volumes
Measuring volumes precisely is an important facet of chemistry, because it impacts the result of experiments and reactions. A small error in measurement can result in incorrect outcomes, making it important to know the assorted strategies for measuring volumes.
These strategies may be broadly categorised into 4 classes: Burettes, Measuring Cylinders, Pipettes, and Syringes. Every methodology has its personal benefits and limitations, which can be mentioned within the following sections.
Burettes
Burettes are units used to measure the quantity of a liquid with excessive precision. They encompass a slender, lengthy tube with a faucet on the backside, which permits for the discharge of the liquid. Burettes can be found in several sizes, starting from 10 mL to 100 mL.
The amount measured by a burette is correct to inside 0.05 mL.
The benefits of burettes embrace excessive precision, ease of use, and the power to precisely measure small volumes. Nevertheless, they are often liable to errors on account of calibration points and dealing with. Burettes are generally utilized in titration experiments, the place a identified quantity of an answer is added to a reactant till the response is full.
Benefits of Burettes:
- Excessive precision
- Ease of use
- Correct measurement of small volumes
Limitations of Burettes:
- Vulnerable to calibration errors
- Dealing with may be difficult
Measuring Cylinders
Measuring cylinders, also called volumetric flasks, are units used to measure the quantity of a liquid with average precision. They encompass a cylindrical tube with markings on the aspect, which point out the quantity measured. Measuring cylinders can be found in several sizes, starting from 10 mL to 1000 mL.
The benefits of measuring cylinders embrace ease of use, average precision, and the power to precisely measure massive volumes. Nevertheless, they are often liable to errors on account of calibration points and the necessity to learn the markings precisely. Measuring cylinders are generally utilized in laboratory settings the place a big quantity of an answer must be measured.
Benefits of Measuring Cylinders:
- Ease of use
- Reasonable precision
- In a position to precisely measure massive volumes
Limitations of Measuring Cylinders:
- Vulnerable to calibration errors
- Ambiguity in markings might lead to errors
Pipettes
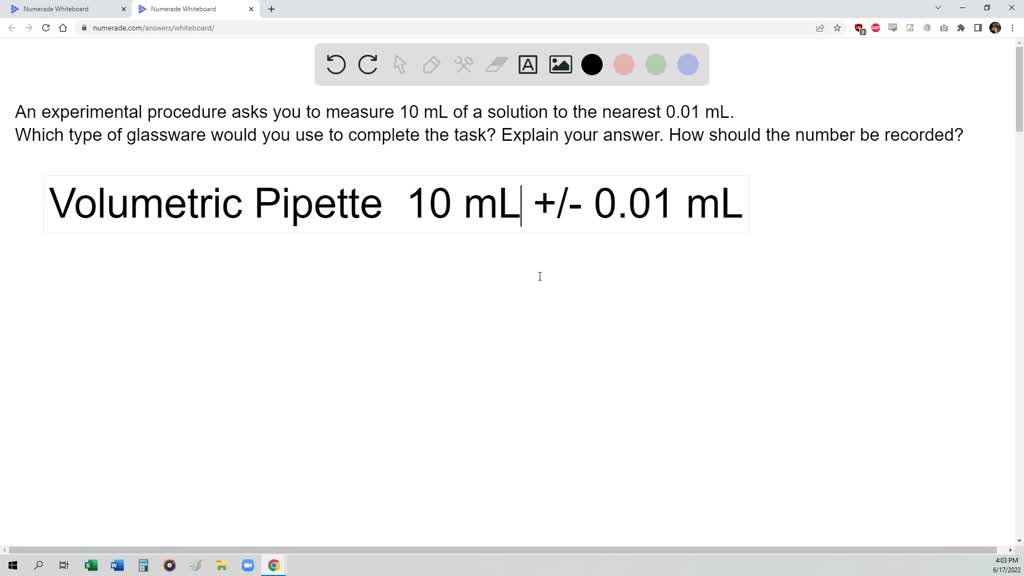
Pipettes are units used to measure the quantity of a liquid with excessive accuracy. They encompass a tube with a bulb on the prime, which permits for the measurement of small volumes. Pipettes can be found in several sizes, starting from 20 μL to 1 mL.
The benefits of pipettes embrace excessive accuracy, ease of use, and the power to precisely measure small volumes. Nevertheless, they are often liable to errors on account of calibration points and the necessity to deal with the pipette rigorously. Pipettes are generally utilized in laboratory settings the place small volumes of an answer must be measured.
Benefits of Pipettes:
- Excessive accuracy
- Ease of use
- Correct measurement of small volumes
Limitations of Pipettes:
- Vulnerable to calibration errors
- Requires cautious dealing with to keep away from errors
Syringes
Syringes are units used to measure the quantity of a liquid with average precision. They encompass a cylindrical tube with a plunger that may be moved to vary the quantity measured. Syringes can be found in several sizes, starting from 1 mL to 10 mL.
The benefits of syringes embrace ease of use, average precision, and the power to precisely measure small to average volumes. Nevertheless, they are often liable to errors on account of calibration points and the necessity to deal with the syringe rigorously. Syringes are generally utilized in laboratory settings the place small to average volumes of an answer must be measured.
Benefits of Syringes:
- Ease of use
- Reasonable precision
- Correct measurement of small to average volumes
Limitations of Syringes:
- Vulnerable to calibration errors
- Ambiguity in markings might lead to errors
| Technique | Precision | Accuracy | Ease of Use | Examples of Purposes |
|---|---|---|---|---|
| Burettes | Excessive | Excessive | Excessive | Titration experiments |
| Measuring Cylinders | Medium | Medium | Excessive | Lab settings the place massive volumes are measured |
| Pipettes | Excessive | Excessive | Excessive | Lab settings the place small volumes are measured |
| Syringes | Medium | Medium | Excessive | Lab settings the place small to average volumes are measured |
Greatest Practices for Measuring 20ml Precisely

Measuring small volumes precisely is essential in chemistry, as even slight variations can impression the result of experiments. To attain constant outcomes, it is important to standardize laboratory tools and methods. On this context, we’ll talk about the most effective practices for measuring 20ml precisely.
Significance of Standardizing Laboratory Gear
Standardizing laboratory tools is important to make sure correct measurements. This entails utilizing tools that has been calibrated and validated to make sure its accuracy.
In line with the Nationwide Institute of Requirements and Know-how (NIST), laboratory tools have to be calibrated no less than yearly to make sure its accuracy.
To attain this, laboratories ought to:
- Use tools that’s calibrated and validated by a certified technician or producer.
- Keep a report of kit calibration and validation to make sure accountability.
- Usually examine tools for indicators of damage and tear, and exchange it as wanted.
Verifying the Accuracy of Measuring Gadgets
Measuring units, comparable to pipettes and burettes, have to be verified for accuracy earlier than use. This entails calibration and validation procedures to make sure that the units are measuring volumes precisely. To confirm the accuracy of measuring units, laboratories ought to: Whereas utilizing a single methodology for measuring 20ml precisely may be efficient, counting on a mix of strategies can present a better diploma of accuracy. This may contain utilizing a number of measuring units to measure the identical quantity, or utilizing a mix of guide and automatic strategies. In conclusion, measuring 20ml precisely is essential in chemistry, and standardizing laboratory tools and verifying the accuracy of measuring units are important to attaining constant outcomes. By following greatest practices and utilizing a mix of strategies, laboratories can guarantee correct measurements and dependable outcomes. Measuring volumes precisely is essential in chemistry to acquire dependable outcomes. Nevertheless, a number of challenges can compromise the accuracy of quantity measurements, affecting the result of experiments and procedures. One of the important frequent challenges in measuring volumes is temperature variations. Temperature fluctuations could cause liquids to broaden or contract, leading to inaccurate measurements. As an example, water expands by roughly 0.1% for each diploma Celsius enhance in temperature. Temperature Results on Liquids: Liquids broaden when heated and contract when cooled. Because of this the quantity of a liquid will enhance with a rise in temperature and reduce with a lower in temperature. Adjusting for Temperature: To beat the consequences of temperature variations, you should use thermometers to measure the temperature of the liquid after which modify the quantity measurements accordingly. You too can use temperature-controlled measuring tools or retailer the liquid in a thermally steady container. Instance: In a titration experiment, a change in temperature can have an effect on the quantity of the titrant and the analyte, resulting in inaccurate outcomes. To mitigate this, use a water tub or a thermostatically managed vessel to keep up a relentless temperature. Floor Pressure Results on Liquids: Floor stress causes liquids to behave in a non-ideal method, resulting in inaccurate measurements. Floor stress could cause liquids to bulge or sag, creating an uneven meniscus. Measuring Strategies: To beat the consequences of floor stress, use specialised measuring methods comparable to utilizing a pipette with a built-in filter or utilizing a syringe with a needle. Instance: In an experiment involving the measurement of the viscosity of a liquid, floor stress can have an effect on the outcomes, resulting in inaccurate conclusions. Meniscus Irregularities: Meniscus irregularities may result from floor stress, contamination, or imperfections within the measuring tools. This may trigger inaccurate quantity measurements. Treatments: To beat meniscus irregularities, use measuring tools with a clean, rounded meniscus. Keep away from utilizing measuring tools with scratches or imperfections. Instance: In a distillation experiment, meniscus irregularities can have an effect on the measurement of the distilled liquid’s quantity, resulting in inaccurate outcomes. Different Components Affecting Quantity Measurements: Different components comparable to contamination, sedimentation, or impurities can even have an effect on quantity measurements. To beat these challenges, use high-quality measuring tools, clear and purify the liquid, and carry out calibration checks. Examples: In an evaluation of a pharmaceutical pattern, impurities or sedimentation can have an effect on the quantity measurement, resulting in inaccurate outcomes. In an environmental monitoring experiment, contamination or sedimentation can have an effect on the measurement, resulting in inaccurate conclusions. Correct measurement in chemistry isn’t just concerning the tools or methods used, but additionally concerning the individuals working them. Correct coaching and high quality management measures are important to make sure that laboratory employees can precisely measure volumes, together with the essential 20ml measurement. Laboratory employees coaching performs a significant function in making certain correct measurements in chemistry. This consists of correct use of kit, calibration procedures, and understanding of the laboratory protocols. Effectively-trained employees are higher geared up to deal with the complexities of laboratory measurements and might determine and proper errors extra successfully. Coaching ought to cowl matters comparable to: Implementing high quality management measures is essential to making sure correct measurements in chemistry. This consists of common tools upkeep, documentation of laboratory procedures, and steady monitoring of laboratory efficiency. High quality management measures ought to embrace:
“Coaching and high quality management are important to making sure correct measurements in chemistry. It isn’t nearly shopping for the precise tools or following procedures, but additionally about growing a tradition of excellence within the laboratory.”
“High quality management measures assist us determine and proper errors earlier than they turn into an issue. It is an ongoing course of that requires dedication and dedication from the whole laboratory crew.” In relation to measuring volumes in a chemistry lab, having the precise tools is essential. The proper tools could make all of the distinction in attaining correct outcomes and making certain the protection of the laboratory. To pick out the precise tools for measuring volumes, a number of components must be thought of. On this part, we’ll talk about the important thing components to contemplate when selecting measuring tools, study examples of specialised tools designed for particular laboratory functions, and clarify the significance of consulting with specialists and laboratory pointers when deciding on tools. When deciding on tools for measuring volumes, precision, accuracy, sturdiness, and person consolation are the important thing components to contemplate. Precision refers back to the tools’s capacity to supply measurements which might be shut to one another. Accuracy refers to how shut the measurements are to the true worth. Sturdiness is a measure of the tools’s capacity to face up to common use with out deteriorating or breaking. Lastly, person consolation is important because it impacts how simply the tools can be utilized and the way comfy the person feels whereas utilizing it. Apart from general-purpose tools, there are specialised devices designed for particular laboratory functions, comparable to titration or chromatography. * Titration tools is designed to measure the quantity of a substance precisely. That is significantly essential in chemistry labs the place exact measurements of quantity are important. As an example, in a chromatography experiment, a laboratory may must measure the quantity of a solvent used to elute a compound. On this case, the selection of kit could be influenced by the necessity for top accuracy and precision. Choosing the proper tools for measuring volumes requires experience and expertise. Consulting with specialists and laboratory pointers is important to make sure that the chosen tools meets the precise wants of the experiment. That is significantly essential when deciding on tools with specialised options or capabilities. By consulting with specialists, laboratories can be certain that the tools they select is correct, exact, sturdy, and user-friendly. Laboratory pointers additionally present useful suggestions for choosing tools that meets particular security requirements. In lots of circumstances, laboratory pointers suggest utilizing tools from respected producers which have a confirmed observe report of manufacturing high-quality merchandise. Laboratories also needs to search for tools that’s calibrated usually to make sure that it stays correct over time. When deciding on tools, it is important to contemplate the precise wants of the experiment, in addition to security and regulatory necessities. Gear calibration is a routine course of that ensures the accuracy and reliability of measurement outcomes. The frequency of calibration depends upon the kind of tools and the precise utility. For instance, a laboratory may calibrate its titration tools each 6 months to make sure that its accuracy and precision meet the necessities of the experiment. Equally, chromatography tools may must be calibrated extra often, comparable to each 3 months, as a result of delicate nature of the experiment. When deciding on tools for calibration, laboratories ought to think about the next components: Measuring volumes precisely is essential in chemistry, and completely different substances current distinctive challenges. Liquids, gases, and solids every require specialised methods and concerns. Correct quantity measurements are important to acquire dependable ends in laboratory procedures and experiments. Liquids are comparatively easy to measure as they comply with the legal guidelines of fluids and are incompressible. Nevertheless, correct measurements are nonetheless essential to keep away from errors. Liquids may be measured utilizing calibrated containers, volumetric flasks, or measuring cylinders. Gases are extremely compressible and their volumes change considerably with temperature and strain. Measuring the quantity of gases requires understanding the properties of gases and utilizing specialised tools comparable to gasoline syringes, burettes, or pipettes. blockquote>Charles’ Legislation: V1 / T1 = V2 / T2 Charles’ Legislation describes the connection between the quantity and temperature of a gasoline, and is important for understanding gasoline properties. Solids pose a singular problem as their quantity may be tough to measure instantly. Solids may be measured by displacing a liquid or utilizing a steadiness to find out their mass after which calculating their quantity primarily based on their density. Measuring volumes precisely requires a deep understanding of the properties of the substances being measured. Density, viscosity, and compressibility are important properties to contemplate when measuring volumes. When measuring volumes in varied substances, it is important to contemplate their properties and use applicable methods and tools to acquire correct outcomes. When working in a laboratory surroundings, it’s essential to prioritize security to stop accidents and make sure the well-being of people concerned. Measuring volumes requires cautious dealing with of kit and substances, and adhering to security protocols is important. On this part, we’ll talk about the significance of security precautions when measuring volumes in a laboratory setting. Private protecting tools (PPE) is a crucial part of laboratory security. It serves as a barrier between the person and doubtlessly hazardous substances or tools. The usage of PPE is required when working with chemical compounds, dealing with tools, or performing procedures that will contain publicity to hazardous supplies. Examples of PPE embrace gloves, goggles, lab coats, and face shields. When deciding on PPE, think about the precise hazards of the substances or tools getting used. As an example, utilizing gloves with chemical resistance when dealing with acids or bases. Moreover, be certain that PPE is correctly fitted and maintained to make sure effectiveness. Accidents can occur even with correct precautions. Having a spill response process in place is essential to minimizing the impression of a spill. This consists of understanding methods to comprise the spill, neutralize the substance (if crucial), and evacuate the world till it’s protected to return. The final steps for spill response embrace: Correct data of laboratory actions and procedures are important for a number of causes. Firstly, they supply a historic context for experiments, permitting researchers to trace progress and determine areas for enchancment. Secondly, they function a vital part of regulatory compliance, making certain that researchers meet trade requirements and rules. Lastly, correct data present useful info for future analysis and growth. Beneficial practices for sustaining correct data embrace: To make sure a protected laboratory surroundings, comply with these really useful security protocols: Measuring 20ml precisely is a talent that requires observe and persistence, however with the precise data and methods, you’ll be able to obtain correct outcomes. Bear in mind, correct measurement is the important thing to dependable outcomes, and with this information, you can be effectively in your method to changing into a proficient chemist. Whether or not you are a pupil or an expert, this information will offer you the data you must measure 20ml precisely and confidently. What’s the significance of correct measurement in chemistry? Correct measurement is essential in chemistry as a result of it determines the reliability of the outcomes obtained in laboratory procedures. Inaccurate measurement can result in deceptive or invalid outcomes, which might have severe penalties in scientific analysis and product growth. What are the frequent challenges in measuring volumes? The frequent challenges in measuring volumes embrace temperature variations, floor stress, and meniscus irregularities. These components can have an effect on the accuracy of the measurement, and it is important to take them into consideration when measuring volumes. How do I choose the precise tools for measuring volumes? To pick out the precise tools for measuring volumes, you must think about the components of precision, accuracy, sturdiness, and person consolation. You also needs to seek the advice of with specialists and laboratory pointers when deciding on tools. What are the protection precautions when measuring volumes? The security precautions when measuring volumes embrace correct use of private protecting tools, spill response procedures, and sustaining correct data of laboratory actions and procedures.
Mixture Technique vs. Counting on a Single Technique
Frequent Challenges in Measuring Volumes
Temperature Variations
Floor Pressure
Meniscus Irregularities, measure 20ml precisely chemistry
Further Concerns
Coaching and High quality Management for Correct Measurement
The Position of Laboratory Employees Coaching
The Significance of High quality Management Measures
Deciding on the Proper Gear for Measuring Volumes

Precision, Accuracy, Sturdiness, and Person Consolation: Key Components to Think about
As an example, a laboratory may must measure the quantity of a substance in a titration experiment. On this case, precision and accuracy are paramount to make sure that the ultimate result’s correct. Nevertheless, sturdiness and person consolation additionally turn into essential concerns when the experiment is carried out repeatedly or over an prolonged interval.
Specialised Measuring Gear for Particular Laboratory Purposes
* Chromatography tools is designed to measure the separation of compounds primarily based on their molecular weights and different properties. That is generally utilized in biochemistry and pharmacology labs.
Consulting with Specialists and Laboratory Tips
Examples of Gear Calibration
Calibration Concerns
Measuring Volumes in Varied Substances
Measuring Volumes in Liquids
Measuring Volumes in Gases
Measuring Volumes in Solids
Understanding Substance Properties
Property
Definition
Density
The mass per unit quantity of a substance.
Viscosity
The measure of a fluid’s resistance to move.
Compressibility
The measure of how a substance’s quantity adjustments with strain.
Security Precautions When Measuring Volumes
Correct Use of Private Protecting Gear
Spill Response Procedures
Sustaining Correct Data of Laboratory Actions and Procedures
Beneficial Security Protocols for Measuring Volumes within the Laboratory
Ultimate Wrap-Up: How To Measure 20ml Precisely Chemistry
Knowledgeable Solutions