Easy methods to learn burette readings correctly, the inspiration of correct analytical chemistry, is an important talent for laboratory professionals to grasp. By understanding the historical past and improvement of burettes, in addition to the correct strategies for calibration, meniscus studying, and temperature management, researchers can guarantee exact leads to chemical reactions and measurements.
The significance of correct burette readings extends past laboratory settings, with purposes in industrial course of management, analysis and improvement, and environmental monitoring. On this article, we’ll delve into the world of burettes, exploring the different sorts, correct utilization, and suggestions for acquiring correct readings.
Easy methods to Use a Burette in Laboratory Measurements
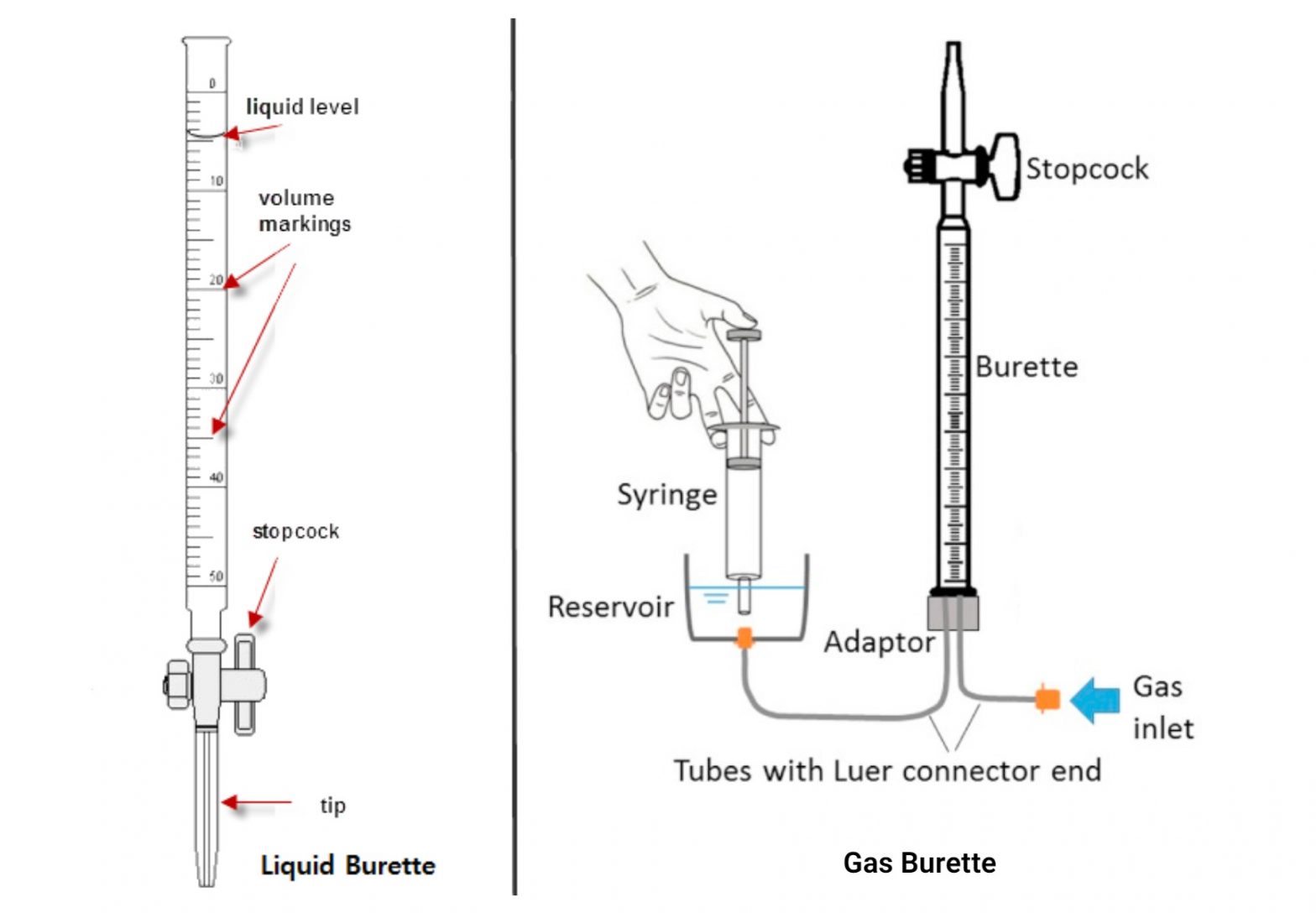
A burette is an important laboratory instrument used for exact measurements of the quantity of liquids, significantly within the context of acid-base titrations. To make sure correct measurements, it’s essential to correctly calibrate the burette and grasp the strategies concerned in utilizing it.
Calibrating the Burette
Calibrating a burette entails verifying its accuracy by evaluating it to a recognized customary. The method begins with zeroing the burette, which entails pouring distilled water into the burette till the extent indicator (the meniscus) rests on the zero mark. It’s important to regulate the burette to room temperature earlier than calibrating, as temperature adjustments can have an effect on the quantity of the liquid. Subsequent, switch a recognized quantity of the liquid right into a container, after which use the burette to measure the identical quantity. Repeat this course of a number of occasions to reduce any errors. Lastly, examine the precise quantity measured by the burette to the recognized customary to confirm its accuracy.
Studying the Meniscus
Studying the meniscus in a burette precisely is essential for acquiring exact measurements. The meniscus is the curve of the liquid’s floor within the burette. To make sure correct readings, make sure the leveling bubble is centered on the meniscus. This ensures the burette is completely degree, which is important for correct measurements. If the leveling bubble will not be centered, modify the burette’s place till it’s. To additional decrease errors, modify the burette to make sure it’s parallel to the sunshine supply. This ensures the meniscus is optically clear and simpler to learn. Lastly, learn the meniscus at eye degree, guaranteeing the studying is taken straight from the meniscus’s curved floor. It is strongly recommended to take a number of readings and common them to scale back any random errors.
Function of the Burette in Acid-Base Titrations
The burette performs a pivotal position in acid-base titrations, the place it’s used to measure the quantity of acid or base added to an answer till a impartial pH is achieved. On this course of, the burette is stuffed with a recognized focus of acid or base, after which transferred to a container containing a recognized quantity of an acid or base with an unknown focus. The acid or base within the burette is then slowly added to the answer till a colour change or pH indicator sign exhibits that the impartial level has been reached. By precisely measuring the quantity of acid or base added, scientists can exactly decide the focus of the unknown answer. This course of is extensively utilized in numerous fields, together with chemistry, biology, and environmental science, to find out the focus of gear in options.
Significance of Correct Measurements
Correct measurements utilizing a burette are essential in numerous laboratory settings. In acid-base titrations, correct measurements permit scientists to exactly decide concentrations, which is important in fields comparable to chemistry and biology. Moreover, exact measurements additionally allow the exact management of reactions, which is crucial in industries the place chemical reactions are an important side, like manufacturing prescription drugs. Moreover, using a burette helps decrease errors, guaranteeing dependable leads to important analysis and improvement processes.
Suggestions for Acquiring Correct Burette Reads
To reduce errors when studying the meniscus from a burette, it’s essential to make use of strategies that promote correct readings. The next methods can assist obtain this purpose.
Minimizing Parallax Error when Studying the Meniscus
When studying the meniscus on a burette, parallax error can result in inaccurate measurements. Nonetheless, this may be minimized by adjusting the viewing angle and the optical readability of the meniscus. One efficient technique is to make use of a vernier scale or a calibrated reticle to align the meniscus with the graduated scale on the burette. This method permits for extra exact willpower of the liquid degree. Moreover, guaranteeing that the viewing space is free from glare and obstructions may scale back parallax error.
- Use a burette with a vernier scale to enhance the accuracy of meniscus readings.
This lets you make exact changes to the viewing angle, minimizing parallax error. - Make use of a calibrated reticle or the same aiming system to make sure exact alignment of the meniscus with the graduate marks on the burette.
This permits correct willpower of the liquid degree, even at low resolutions. - Keep away from making readings in areas with direct mild or reflections that may intervene with the viewing expertise.
This may be achieved by positioning the burette close to a lightweight supply however in a roundabout way in its path, or by utilizing a defend to diffuse the sunshine. - Use a burette with a transparent, unobstructed view of the meniscus, permitting for straightforward identification of the liquid degree.
Controlling Temperature and Environmental Components
Temperature can considerably have an effect on the readings obtained from a burette. When taking measurements, it’s important to manage the temperature of the burette and the encircling setting to stop errors.
Temperature Management
The temperature of the burette and the encircling setting must be stored steady to stop errors attributable to thermal growth. If doable, use a temperature-controlled setting or a heating/cooling system to manage the temperature.
Temperature must be maintained inside 2-5 levels Celsius of the specified temperature to stop vital errors.
Environmental Management
Along with temperature management, the encircling setting also needs to be managed to stop errors attributable to adjustments in humidity, air strain, or different environmental elements.
Exact readings require a managed setting with minimal adjustments in temperature, humidity, and air strain.
Protected Dealing with and Upkeep of Burettes, Easy methods to learn burette studying
Burettes must be dealt with and maintained safely to make sure optimum efficiency and longevity. Cleansing and storage procedures can assist stop contamination and harm to the gear.
Cleansing and Storage Procedures
1. Rinse the burette totally with distilled water after every use to stop contamination.
2. Use delicate cleaning soap and keep away from abrasive supplies to scrub the burette.
3. Rinse the burette with distilled water after cleansing to take away any cleaning soap residue.
4. Dry the burette fully earlier than storing it.
5. Retailer the burette in a protecting case or container to stop harm.
Preventive Upkeep
Common upkeep can assist stop harm to the burette and guarantee optimum efficiency.
- Examine the burette for indicators of injury or put on frequently.
Examine the graduate marks, the stopcock, and the physique of the burette for any indicators of injury or put on. - Clear and lubricate the stopcock commonly to make sure easy operation.
- Carry out a calibration test on the burette periodically to make sure accuracy.
This entails checking the quantity of the burette towards a licensed customary.
Utilizing Burette Readings in Calculations: How To Learn Burette Studying
Utilizing burette readings in calculations is an important step in laboratory measurements, as they supply correct information for figuring out the focus of options and the quantity of substance added. This course of entails utilizing numerous calculations, together with molarity and normality, to acquire dependable outcomes.
To start, it is important to know the several types of calculations concerned in utilizing burette readings. Molarity, as an illustration, is a measure of the focus of an answer in moles of solute per liter of answer, sometimes denoted by the image ‘M’. Normality, however, is a measure of the focus of an answer in equal weights of solute per liter of answer, represented by the image ‘N’.
Molarity Calculations
Molarity calculations are elementary to figuring out the focus of an answer utilizing a burette. The method for molarity is given by:
molarity (M) = (moles of solute) / (liters of answer)
To calculate the molarity of an answer, begin by measuring the quantity of the answer utilizing the burette, then proceed to weigh or measure the quantity of solute added. For instance, for example you are working with a burette studying of 25 mL of an answer containing 0.25 g of sodium chloride (NaCl), which has a molar mass of 58.44 g/mol. To calculate the molarity of the answer, you should utilize the next steps:
- First, decide the variety of moles of NaCl added:
- Subsequent, calculate the quantity of the answer in liters:
- Lastly, calculate the molarity of the answer:
moles NaCl = mass NaCl / molar mass NaCl = 0.25 g / 58.44 g/mol = 0.0043 mol
quantity (L) = 25 mL / 1000 = 0.025 L
molarity (M) = moles NaCl / quantity (L) = 0.0043 mol / 0.025 L = 0.172 M
Normality Calculations
Normality calculations are important for figuring out the focus of an answer in equal weights of solute. The method for normality is given by:
normality (N) = (equal weight of solute) / (liters of answer)
To calculate the normality of an answer, begin by measuring the quantity of the answer utilizing the burette, then proceed to weigh or measure the quantity of solute added. For instance, for example you are working with a burette studying of 25 mL of an answer containing 0.25 g of sodium hydroxide (NaOH), which has an equal weight of 40 g/equiv. To calculate the normality of the answer, you should utilize the next steps:
- First, decide the variety of moles of NaOH added:
- Subsequent, calculate the full cost (in equivalents) of the solute:
- Lastly, calculate the normality of the answer:
moles NaOH = mass NaOH / molar mass NaOH = 0.25 g / 40 g/mol = 0.00625 mol
whole cost = moles NaOH x equal weight = 0.00625 mol x 1 equiv = 0.00625 equiv
normality (N) = whole cost / quantity (L) = 0.00625 equiv / 0.025 L = 0.25 N
Stoichiometric Calculations
Along with molarity and normality calculations, stoichiometric calculations additionally play a pivotal position in figuring out the quantity of substance added utilizing a burette. Stoichiometry is the examine of the quantitative relationships between reactants and merchandise in chemical reactions.
To carry out stoichiometric calculations, we have to decide the mole ratio of the reactants and merchandise concerned within the response. That is sometimes performed by balancing the chemical equation for the response.
For instance, let’s contemplate a response between sodium hydroxide (NaOH) and hydrochloric acid (HCl):
NaOH + HCl -> NaCl + H2O
The balanced equation is:
2NaOH + HCl -> 2NaCl + H2O
To find out the quantity of substance added utilizing a burette, we have to know the mole ratio between NaOH and HCl. From the balanced equation, we are able to see that 2 moles of NaOH react with 1 mole of HCl.
If we’re working with a burette studying of 25 mL of an answer containing 0.25 g of NaOH, we are able to calculate the variety of moles of NaOH added utilizing the next steps:
* decide the variety of moles of NaOH added
* calculate the quantity of the answer in liters
* calculate the mole ratio between NaOH and HCl
* decide the quantity of HCl required to react with the quantity of NaOH added
The significance of precisely changing between models of measurement when working with burettes can’t be overstated. Standardization procedures are important for guaranteeing the accuracy of the info obtained utilizing a burette.
When working with burettes, it is essential to know the ideas of molarity, normality, and stoichiometry. By mastering these calculations, scientists can precisely decide the focus of options and the quantity of substance added utilizing a burette.
The relevance of standardization procedures can’t be overstated. Standardization entails calibrating the burette to make sure that the measured quantity is correct and constant. This entails measuring the quantity of a recognized answer utilizing the burette and evaluating it to the anticipated worth.
The significance of precisely changing between models of measurement when working with burettes lies in the truth that the info obtained utilizing a burette is used to make important selections in scientific analysis and industrial purposes.
The applying of burette readings in figuring out the stoichiometry of chemical reactions is crucial for understanding the quantitative relationships between reactants and merchandise.
In conclusion, using burette readings in calculations entails understanding the ideas of molarity, normality, and stoichiometry. Standardization procedures are important for guaranteeing the accuracy of the info obtained utilizing a burette. The significance of precisely changing between models of measurement when working with burettes can’t be overstated.
Superior Purposes of Burette Readings

Along with its conventional use in laboratory measurements, the burette has discovered purposes in numerous superior analytical strategies, course of management, and analysis and improvement. This consists of using burettes in chromatography and spectroscopy, course of management and monitoring in industrial settings, and analysis and improvement for testing and validating new analytical strategies.
The Use of Burette Readings in Chromatography and Spectroscopy
Burette readings play an important position in chromatography and spectroscopy, two superior analytical strategies used to investigate and establish the composition of gear. Chromatography separates the elements of a combination primarily based on their chemical properties, whereas spectroscopy measures the interplay between matter and electromagnetic radiation. Correct burette readings are important in these strategies as they permit researchers to find out the focus of gear in a pattern, which is important in figuring out and quantifying their composition.
Burette readings are utilized in numerous varieties of chromatography, together with fuel chromatography (GC), liquid chromatography (LC), and supercritical fluid chromatography (SFC). In GC, burette readings are used to calculate the quantity of substance injected into the chromatograph, whereas in LC, burette readings are used to find out the focus of the cell part. SFC makes use of a supercritical fluid because the cell part, and burette readings are used to manage the strain and temperature of the system.
Spectroscopy makes use of numerous strategies to measure the interplay between matter and electromagnetic radiation, together with infrared (IR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and mass spectrometry (MS). Burette readings are utilized in these strategies to find out the focus of gear in a pattern, which is important in figuring out and quantifying their composition.
The Function of Burette Readings in Course of Management and Monitoring
Burette readings play an important position in course of management and monitoring in industrial settings. Course of management entails the measurement and management of course of variables comparable to temperature, strain, and movement fee to make sure that a course of is operating easily and effectively. Monitoring entails the continual measurement of course of variables to detect any adjustments or abnormalities.
Burette readings are utilized in numerous industrial processes, together with chemical processing, meals processing, and pharmaceutical manufacturing. In chemical processing, burette readings are used to manage the focus of reactants and merchandise in a chemical response. In meals processing, burette readings are used to watch the pH and temperature of meals merchandise throughout processing. In pharmaceutical manufacturing, burette readings are used to manage the focus of lively components in a closing product.
Actual-time measurements and information evaluation are important in course of management and monitoring. Burette readings allow researchers to watch course of variables in real-time and make changes as wanted to make sure that the method is operating easily and effectively. That is important in stopping gear failure, product contamination, and different high quality management points.
The Utility of Burette Readings in Analysis and Improvement
Burette readings are utilized in analysis and improvement for testing and validating new analytical strategies. Researchers use burette readings to find out the accuracy and precision of recent analytical strategies, which is important in guaranteeing that the strategy is dependable and reproducible.
Burette readings are utilized in numerous analysis areas, together with environmental science, organic science, and supplies science. In environmental science, burette readings are used to watch the focus of pollution in water and air samples. In organic science, burette readings are used to find out the focus of antibodies and antigens in a pattern. In supplies science, burette readings are used to manage the focus of reactants and merchandise in a chemical response.
Burette readings allow researchers to check and validate new analytical strategies, which is important in guaranteeing that the strategy is correct and dependable. That is important in stopping errors in information interpretation and guaranteeing that analysis outcomes are dependable and reproducible.
Finest Practices for Burette Upkeep and Security
Common upkeep and calibration of burettes are essential for guaranteeing optimum efficiency and accuracy in laboratory measurements. A well-maintained burette gives dependable outcomes, reduces the danger of errors, and prolongs the instrument’s lifespan. On this part, we’ll focus on the significance of standard upkeep and calibration, the hazards related to burettes, and finest practices for disposing of and recycling them.
Significance of Common Upkeep and Calibration
- Common calibration checks the burette’s accuracy and ensures that it’s free from any blockages or harm.
- Upkeep entails cleansing the burette’s internal surfaces, alternative of worn-out components, and guaranteeing correct functioning.
- Common upkeep and calibration assist stop errors and contamination, which might compromise the integrity of experimental outcomes.
Correct calibration and upkeep procedures are important to make sure that the burette gives correct and dependable readings. This consists of checking for any blockages, leaks, or harm, and cleansing the internal surfaces to take away any residual chemical compounds or particles. Common upkeep additionally entails changing worn-out components and guaranteeing correct functioning of the valve and different elements.
Hazards Related to Burette
The dealing with and use of burettes contain particular hazards that have to be addressed to make sure laboratory security. A few of the hazards related to burettes embrace:
- Chemical Publicity: Burettes might comprise chemical compounds that may trigger pores and skin irritation, harm to eyes, and even respiratory issues.
- Bodily Damage: Improper dealing with or dropping of burettes can result in bodily damage, together with cuts, bruises, or harm to joints.
- Hazardous Waste: Burettes might require disposal and recycling procedures that deal with hazardous waste.
When dealing with burettes, it’s important to take needed precautions to stop chemical publicity and bodily damage. This consists of sporting private protecting gear (PPE), comparable to gloves, security glasses, and lab coats. Correct storage and disposal of burettes are additionally important to stop accidents and guarantee compliance with rules.
Finest Practices for Disposing of and Recycling Burette
Correct disposal and recycling of burettes require cautious consideration to stop environmental harm and guarantee compliance with rules. Some finest practices for disposing of and recycling burettes embrace:
- Accountable Disposal: Get rid of burettes in response to the producer’s directions and native rules.
- Recycling: Take into account recycling burettes to reduce waste and preserve sources.
- Correct Storage: Retailer burettes in a safe and designated space to stop accidents and guarantee accessibility.
To get rid of burettes responsibly, it’s important to observe the producer’s directions and native rules. Recycling burettes additionally helps decrease waste and preserve sources. Correct storage of burettes in a safe and designated space prevents accidents and ensures accessibility.
Final Level

In conclusion, studying burette readings correctly is a important talent that requires consideration to element, understanding of laboratory rules, and adherence to finest practices. By following the strategies and suggestions Artikeld on this article, researchers can guarantee correct and dependable outcomes, advancing our understanding of the chemical world and informing real-world purposes.
Q&A
What’s the significance of correct burette readings in laboratory settings?
Correct burette readings are essential in laboratory settings as they straight affect the accuracy of chemical measurements and reactions. Even small errors can result in incorrect outcomes, which might have vital penalties in analysis and improvement.
How will you decrease parallax error when studying the meniscus in a burette?
To reduce parallax error, use a vernier scale, modify for optical readability, and guarantee good lighting within the laboratory. This may assist you get hold of correct measurements and scale back errors.
What are the hazards related to burettes, and how are you going to deal with them safely?
Burettes can pose chemical publicity and bodily damage hazards. Deal with them with care, observe correct calibration and upkeep procedures, and get rid of them correctly when they’re not wanted.
Can burette readings be utilized in industrial course of management and monitoring?
Sure, burette readings can be utilized in industrial course of management and monitoring. They supply real-time measurements and information evaluation, enabling firms to optimize their manufacturing processes and enhance effectivity.